Warning: This piece mentions suicide.
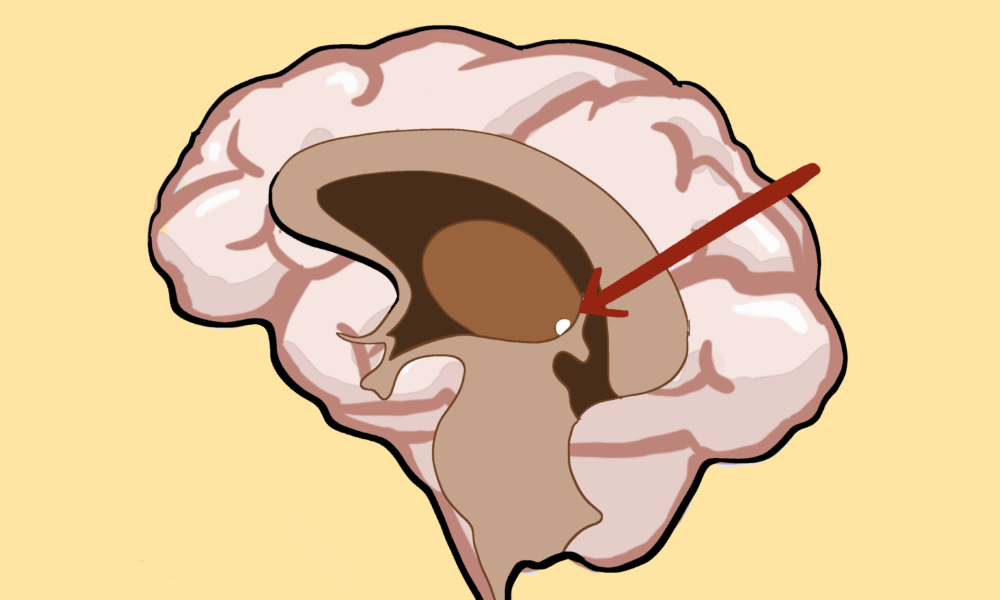
The habenula is a tiny structure buried deep within the brain, composed of two distinct subregions—the medial (MHb) and lateral habenula (LHb). Although small, it is a critical hub for regulating mood, motivation, and reward processing. Because of this, researchers have begun to link abnormalities in this region to major depressive disorder (MDD), a condition that causes persistent feelings of sadness, hopelessness, and a general loss of interest in pleasurable activities.
In a recent review paper in Nature’s Translational Psychiatry, Feiteng Lin, a PhD student in McGill’s Department of Human Genetics and member of Dr. Gustavo Turecki’s Lab at the Douglas Research Centre, synthesized recent research exploring the habenula’s wiring in the brain and how it shapes the pathophysiology of MDD. In an interview with The Tribune, Lin described his deeply personal motivation to study MDD.
“I’m personally interested in [researching] depression because one of my family members [died] by suicide,” Lin said.
Lin highlighted that a major research gap in the field is the limited ability to study MDD-related brain circuitry in living human subjects, explaining that such studies can only be conducted on individuals who have died and donated their brains for research.
“The majority of research has only been done in animal models of depression […] so the major gap I wanted to address is the [lack of] research conducted in humans,” Lin said.
The review found that the two sub-regions of the habenula are affected differently in the brains of MDD patients. Specifically, research on the MHb shows that certain signalling systems appear downregulated, while in the LHb, several stress-related pathways seem abnormally amplified.
Lin also explained how patients who experience prolonged stress tend to have worse symptoms and, therefore, outcomes, which may be related to alterations in the habenula’s neurocircuitry.
“Patients who are generally experiencing stress and anxiety, especially [for longer periods], may develop an overactive habenula,” Lin explained. “My suggestion would be to [develop strategies to decrease anxiety levels] and do more exercise to keep the mood [elevated].”
While ketamine was first used as a surgical anesthetic in the ‘60s, studies from the 2000s proved it was a viable treatment for depression, demonstrating its rapid-acting effects by slowing down the activity of the habenula. Its mechanism blocks the burst firing of LHb neurons, which is significant because abnormal firing is associated with depression-like states.
What makes Lin’s study different from the rest is his focus on the habenula—a brain region that is notoriously difficult to study due to the careful precision required to extract it.
“It’s extremely small, which also makes it difficult to [isolate], and it’s different from other studies using other regions of the brain, such as the prefrontal cortex, which is a large brain tissue [making it] easier to handle,” Lin said. “[Overall], the habenula requires extra carefulness.”
Given that the World Health Organization projects that MDD will become the greatest global burden of disease by 2030, it is imperative that researchers continue investigating its underlying mechanisms and potential treatments. Lin’s study is a promising step in that direction, suggesting that the habenula may play a larger role in MDD than previously understood.“Depression is a leading cause of disability, and it’s affecting lots of people, with many currently [unable to obtain treatment], and therefore continuing to suffer,” Lin said. “There’s multiple avenues towards [investigating the underlying] mechanism of MDD, and so we must keep trying to combine different technologies and strategies [to improve the lives of MDD patients].”
A previous version of this article stated that there are significant changes to the nicotinic acetylcholine receptor in the habenula among those who smoke, quoting Feiteng Lin. The Tribune regrets this error.